Why last week’s consultation response is concerning for patients, the health system and the UK
Once again, the financial pressures facing the NHS reared their ugly head.
NHS England (NHSE) and the National Institute for Clinical Excellence (NICE) made their recommendations in response to their joint consultation into the way drugs are evaluated and funded.
Whilst some concessions have been made, and NICE and NHSE are busy putting forward the line that ‘patients are set to benefit from more rapid access to the most cost-effective treatments.’ But there can be little doubt that today’s outcome should be of concern to patients, the health system and the UK.
Let us demonstrate why.
The £20 million ‘budget impact threshold’ for new technologies originally proposed in the consultation and widely viewed as a potential cap on treatments, has been rebranded as ‘a budget impact test.’
Under this ‘test’, new treatments costing the NHS £20 million per year or more for the first three years will trigger commercial discussions between the manufacturer and NHS England.
This is not an automatic cap on treatments or budget, the likes we have seen, for instance, with hepatitis C treatments. NHSE has stated it will fund new treatments that cost above this threshold.
But the principle is similar. When the Hepatitis C Trust was unsuccessful in its judicial review application against NHS England’s cap on treatments, NHS England stated it “will now focus on securing an improved deal for patients from pharmaceutical companies…”
Should a commercial agreement not be found, NHS England will be able to apply to NICE to phase the introduction of a new therapy, normally for up to three years but longer in exceptional circumstances, so as to manage the affordability impact on the NHS.
This also means their negotiating hand will be much stronger. Whatever NHS England is prepared to pay will likely be more attractive than limited or phased market entry.
Worse still, protracted negotiations will slow down patient access to new, innovative and potentially lifesaving treatments.
There is also a capacity issue here when the test is triggered. The cost threshold is likely to encompass one in every five new treatments that become available. NICE simply does not have the people-power to work through such a large number of complex negotiations.
Again, this will only serve to slow down patient access to treatments, rather than accelerate it.
Are the industrial strategy and Accelerated Access Review now irrelevant?
Against any backdrop, let alone the one of financial uncertainty and pay restraint in which we currently sit, Britain’s life sciences sector is a success story.
Just a month ago, The Times claimed it had seen a government note on which industries are being prioritised in Brexit negotiations: life sciences was ‘high priority.’
It was singled out in the Industrial Strategy White paper.
And what’s more, the Accelerated Access Review talked about getting ground-breaking treatments to patients in need, at a faster pace. Its aim was to meet the challenges of the future.
But the risk is that life sciences will look at this consultation outcome and think:
“Protracted negotiations”
“Pushed down prices”
“Longer time period to get therapies to patients”
“Automatic funding only for curative treatments*”
In short, the UK becomes a much less attractive place for life sciences to pioneer and launch new therapies, and it is patients, the health system and the UK as a whole that will be worse off as a result.
Fast forward and the UK is no longer considered a launch market, UK patients don’t get access to first in class treatments, there are fewer comparator medicines available for clinical trials, and the associated R&D talent and brainpower slowly drifts away.
The UK’s reputation as a place for investing in life sciences will not be helped by the recent controversies about the PPRS rebate. A government that only recently cajoled the industry into putting an additional £250 million into the NHSE budget beyond what had already been agreed will not necessarily be seen as “open for business”.
If approved by the NICE Board, the outcomes will come into force from 1 April this year, with the impact being reviewed in three years’ time.
By that time we will likely already have left Europe.
What then? The UK out of EMA, the EMA out of the UK…..See the latest from Victoria Dean, Partner for Brexit, on the impact our departure from EU will have on the pharmaceutical industry.
The impact on people living with rare conditions (A perspective from Josie Godfrey, Former Assistant Director, NICE Highly Specialised Technologies)
Many will welcome the decision to remove any role for NHSE’s Clinical Priorities Advisory Group (CPAG) from decisions about ultra-orphan drugs and away from the proposed fixed cost per QALY threshold of £100,000 for the products evaluated through the Highly Specialised Technologies programme.
The revised pans will introduce discretion for the HST Committee to recommend products at up to £300,000 per QALY. This goes some way to reward the most effective treatments for very rare diseases. Companies developing curative treatments with a strong evidence base should feel confident that there is an objective, systematic and transparent route to access.
However, for many patients and companies working to support those with very rare conditions this is less good news. The revised approach still overlooks the significant methodological challenges inherent in assessing high-cost, low-volume treatments with a limited evidence-base.
This also signals a move to abandon the criteria that NICE put in place, in part due to the difficulty in generating a reliable cost per quality-adjusted life year (QALY) on the basis of limited data, about both the disease and the treatment. Indeed, NICE initially avoided publishing cost per QALY figures for HST evaluations.
Even if these methodological hurdles can be overcome it is not clear how the incremental QALY gains and associated cost per QALY thresholds have been determined. Whilst 3 of the 4 products that have been through an HST evaluation would benefit from some discretion in the cost per QALY threshold, it is not clear that this discretion would have gone far enough to lead to a positive recommendation. Whilst most share the view that the affordability of new treatments cannot be ignored, it is concerning that no assessment of the likely impact of this new process has been shared.
If you’d like to speak to the Portland Health team about what it means for you, then contact us here.
*The £100,000 QALY threshold for Highly Specialised Technology (HST) evaluations is to be maintained. But above this threshold, the HST Evaluation Committee will have ‘discretion to apply the QALY weight in defined circumstances.’ This will be weighted as follows:
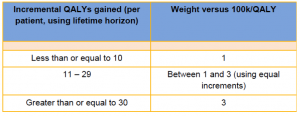
Source, NICE Board Papers, 15.03.17, Available here: https://www.nice.org.uk/Media/Default/Get-involved/Meetings-In-Public/Public-board-meetings/agenda-and-papers-mar-17.pdf
For rare diseases, in particular, the £100,000 QALY threshold will likely be met by only the most exceptional, and perhaps only curative, treatments.

